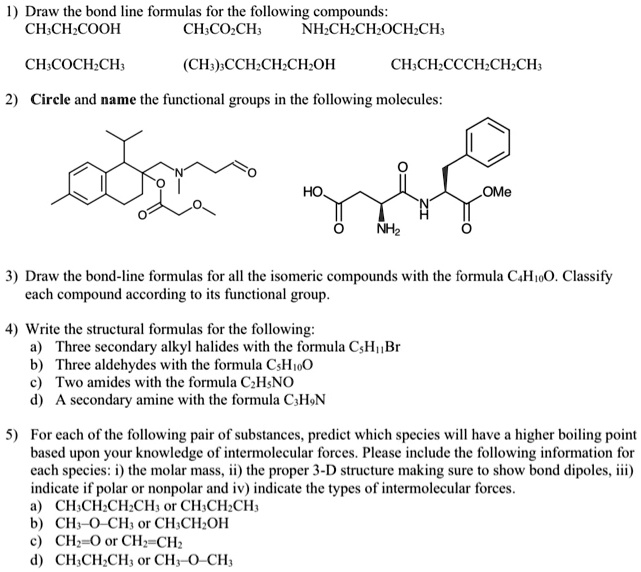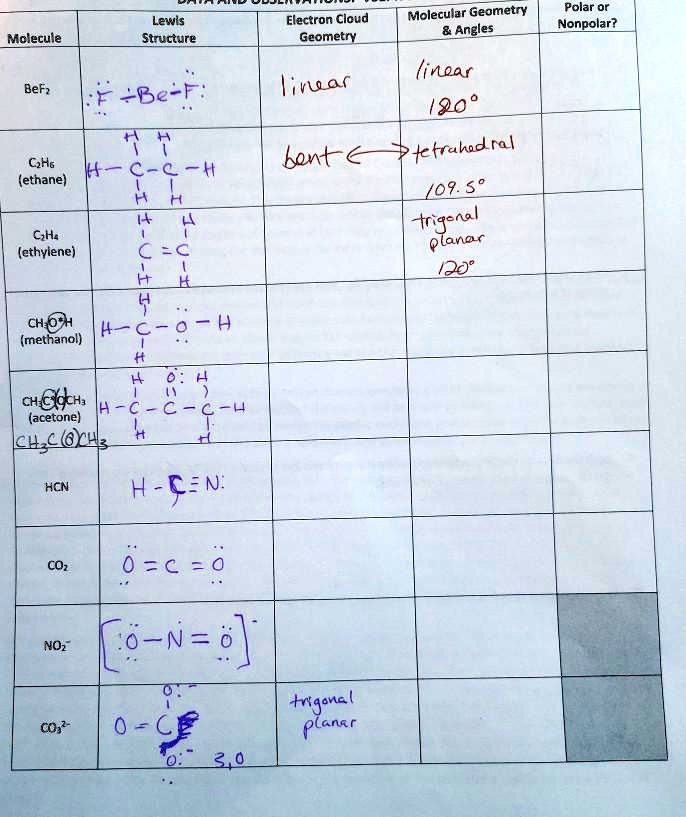
SOLVED: Polar or Nonpolar? Lewis Structure Electron Cloud Geometry Molecular Geometry Angles Molecule Linear 90 Trigonal Bipyramidal 109.5 Tetrahedral 122.8 BeF2 Iodine CH4 (methane) Bond C2H4 (ethylene) CH3OH (methanol) CH3COCH3 (acetone) HCN
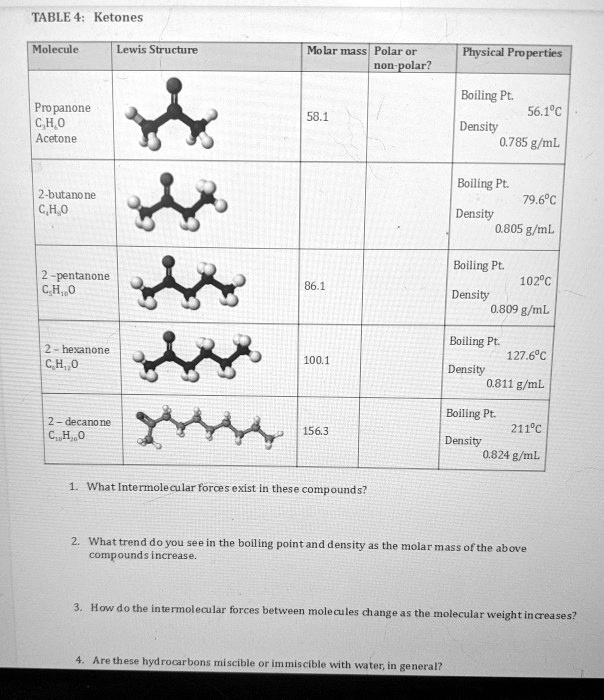
SOLVED: TABLE 4: Ketones Molecule Lewis Structure Molar mass Polar or nonpolar? Physical Properties Boiling Point: 56.1°C Density: 0.785 g/mL Propanone CH3COCH3 Acetone Molar mass: 58.1 Boiling Point: 79.6°C Density: 0.805 g/mL
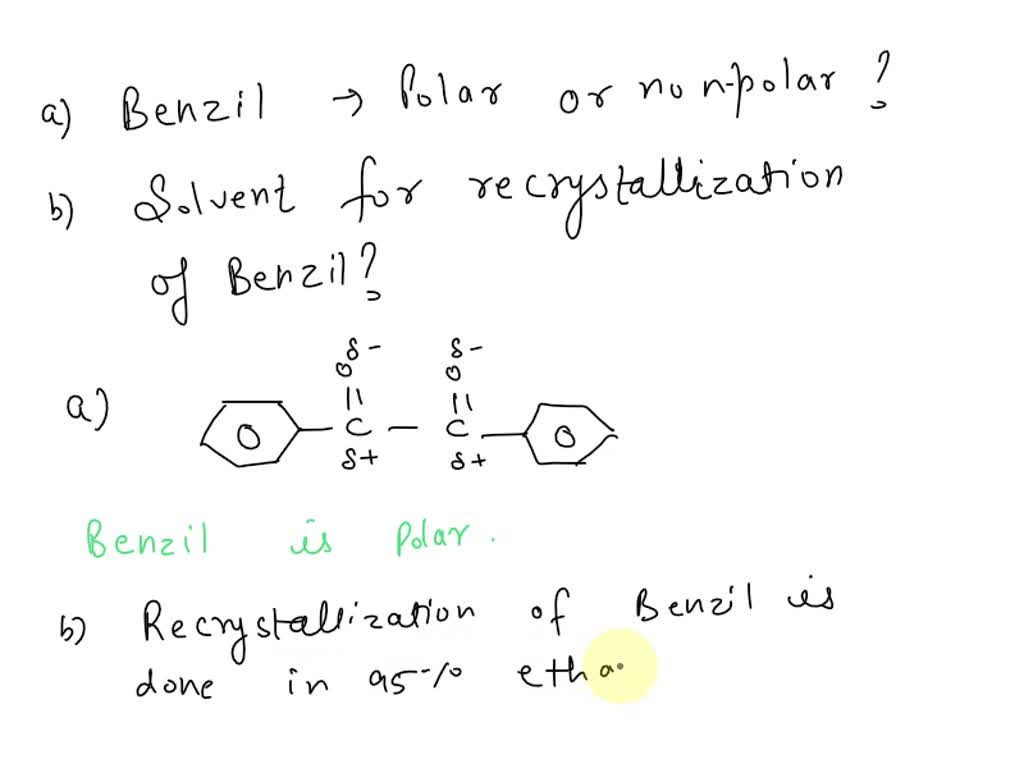
SOLVED: Is Benzil mostly polar, has some polar features, or non-polar? What solvent would be best to use for recrystallization: hexane, acetone, ethanol, or water? Why?

Determine whether the molecule CH_2CHCH_3 is polar: A) The molecule is polar only because there are slightly polar bonds. B) The molecule is polar because there are slightly polar bonds and the
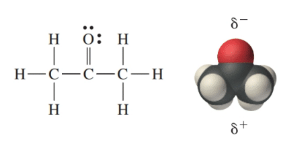
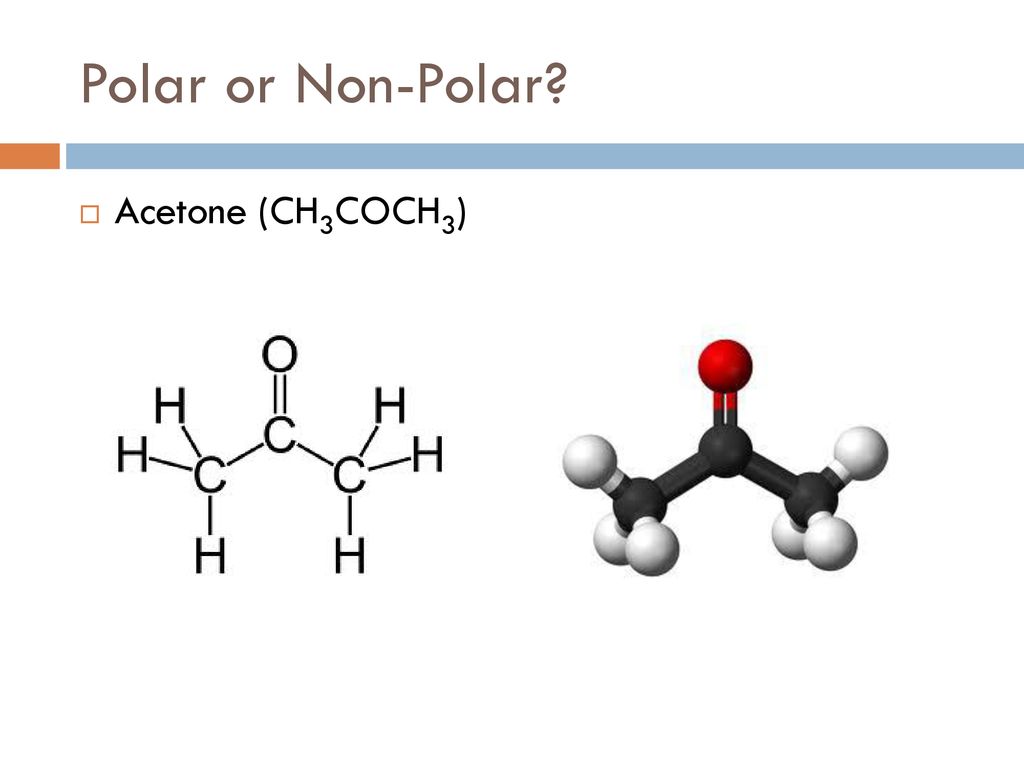
CH 3 COCH 3 (acetone) is a common laboratory solvent that is often used in nail polish remover. Its Lewis structure and space-filling molecular image are shown here: Acetone is a polar