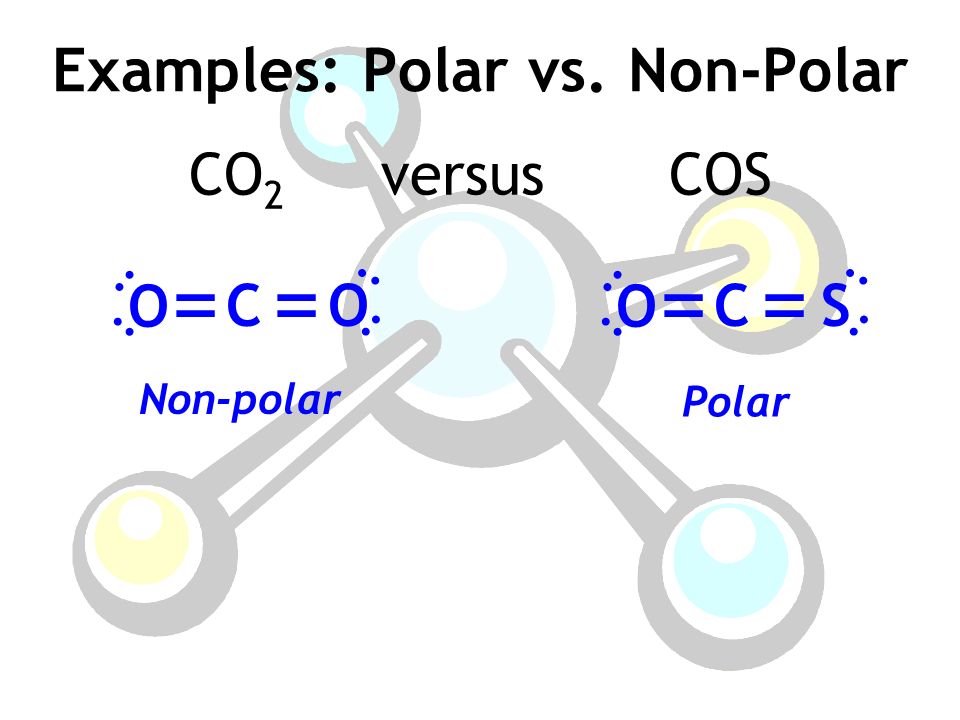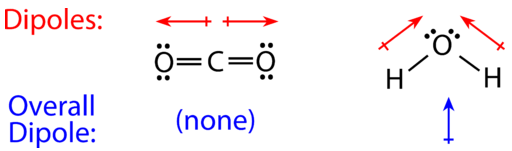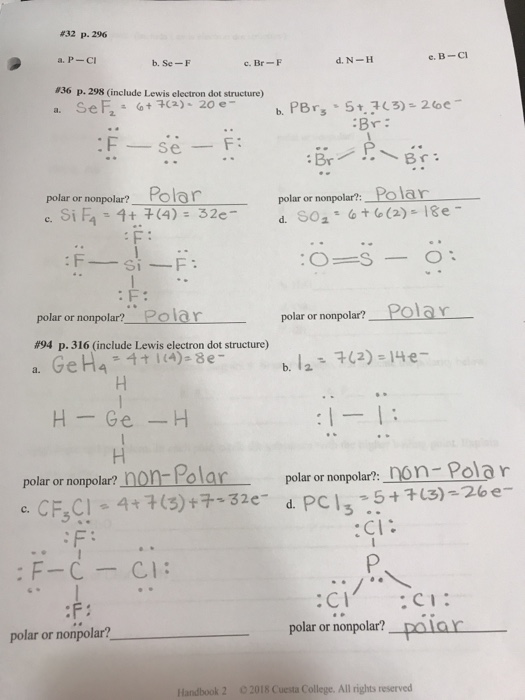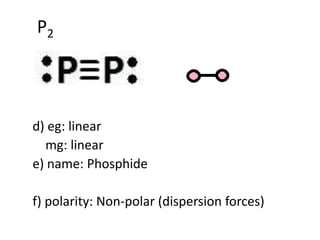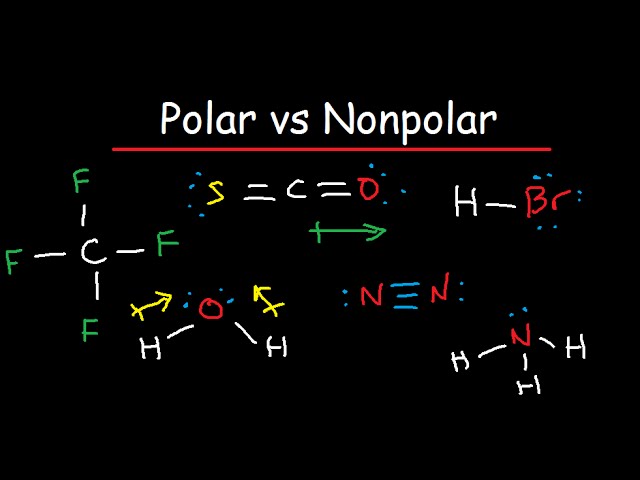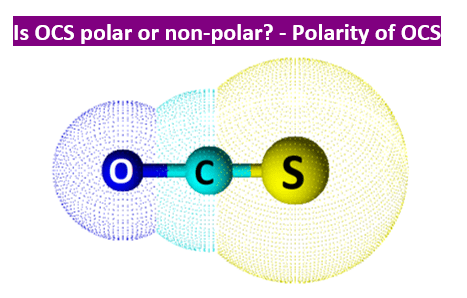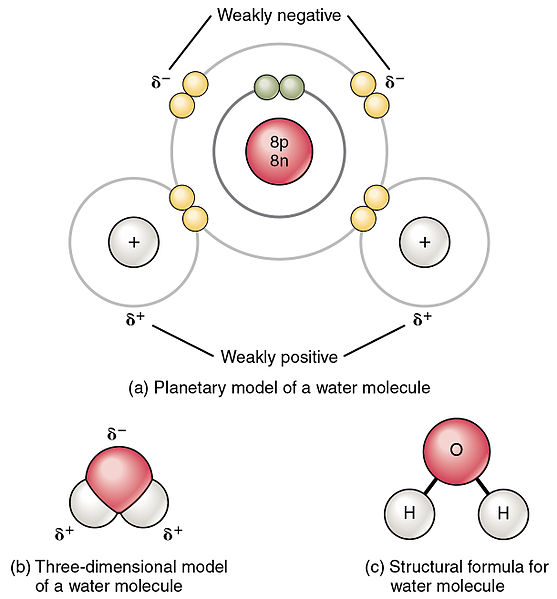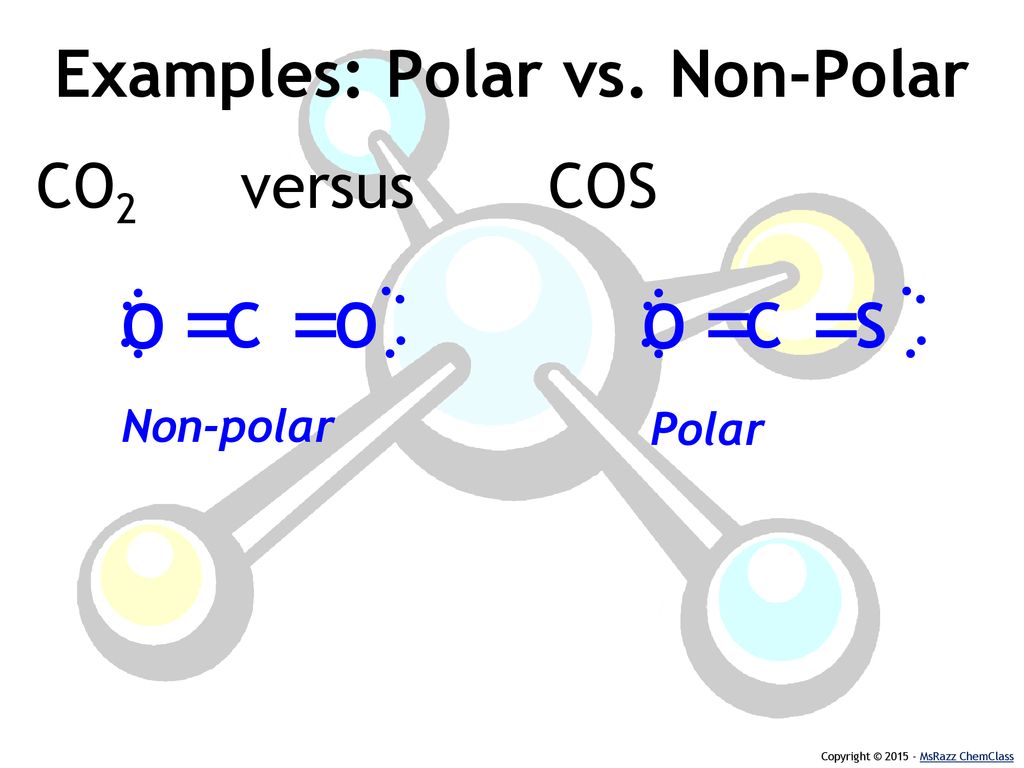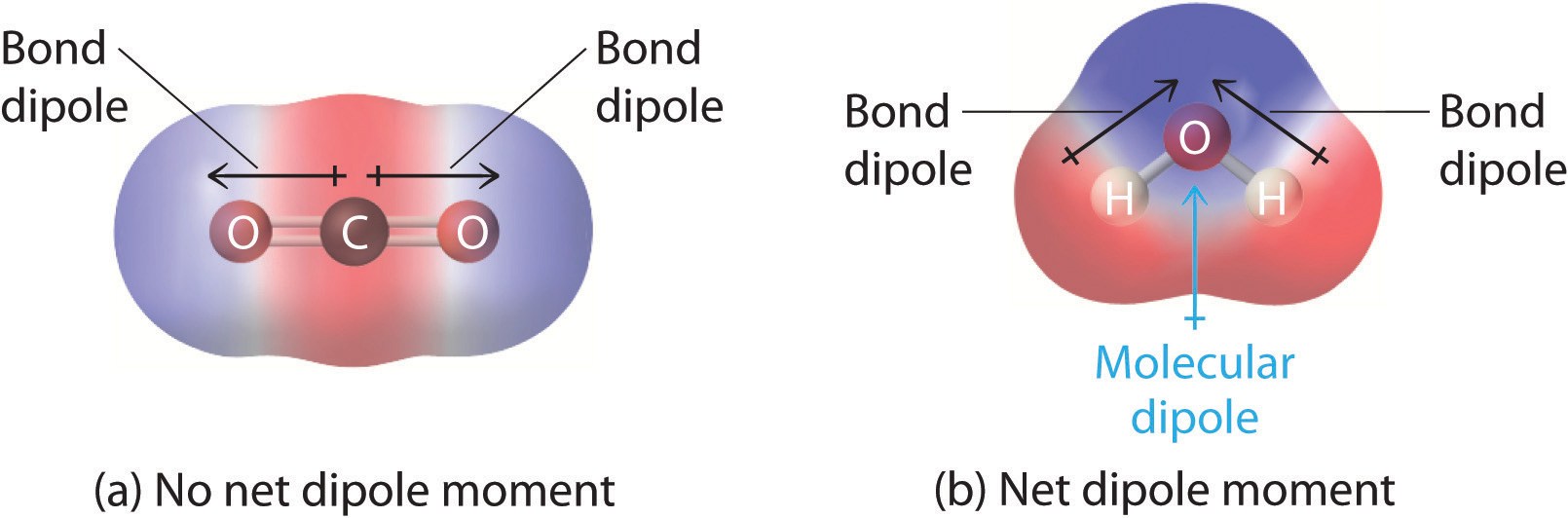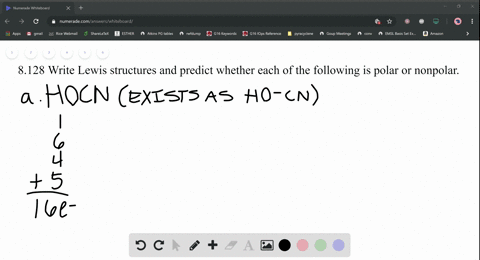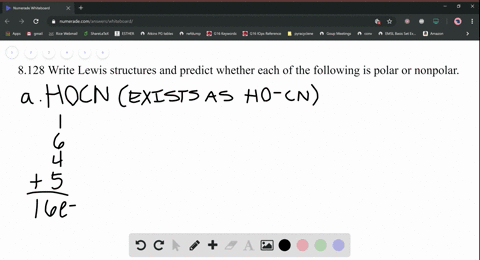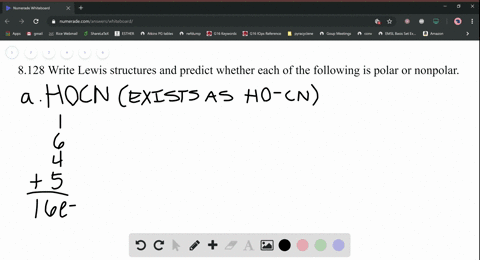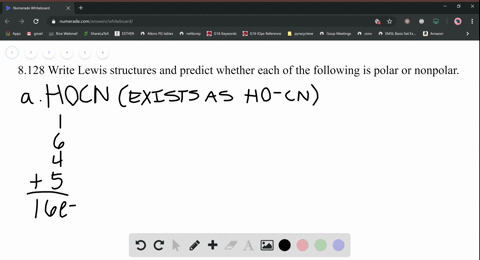
SOLVED: Write Lewis structures and predict whether each of the following is polar or nonpolar: HOCN (exists as HO-CN), COS, XeF2, CF2Cl2, SeF6, H2CO (C is the central atom).

SOLVED: On your Calculations page, draw a Lewis structure for COS and answer the following questions: a. What is the electron group geometry of COS? b. What is the molecular geometry of