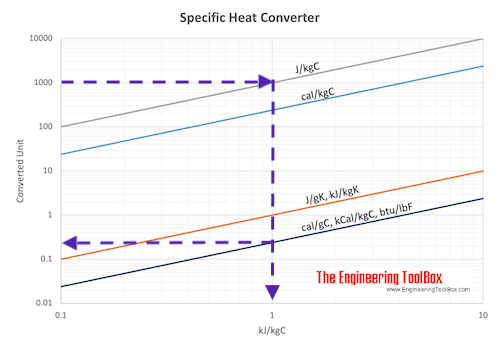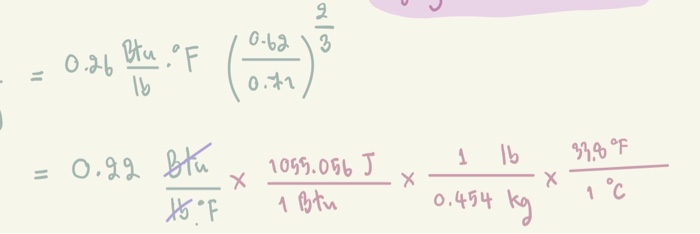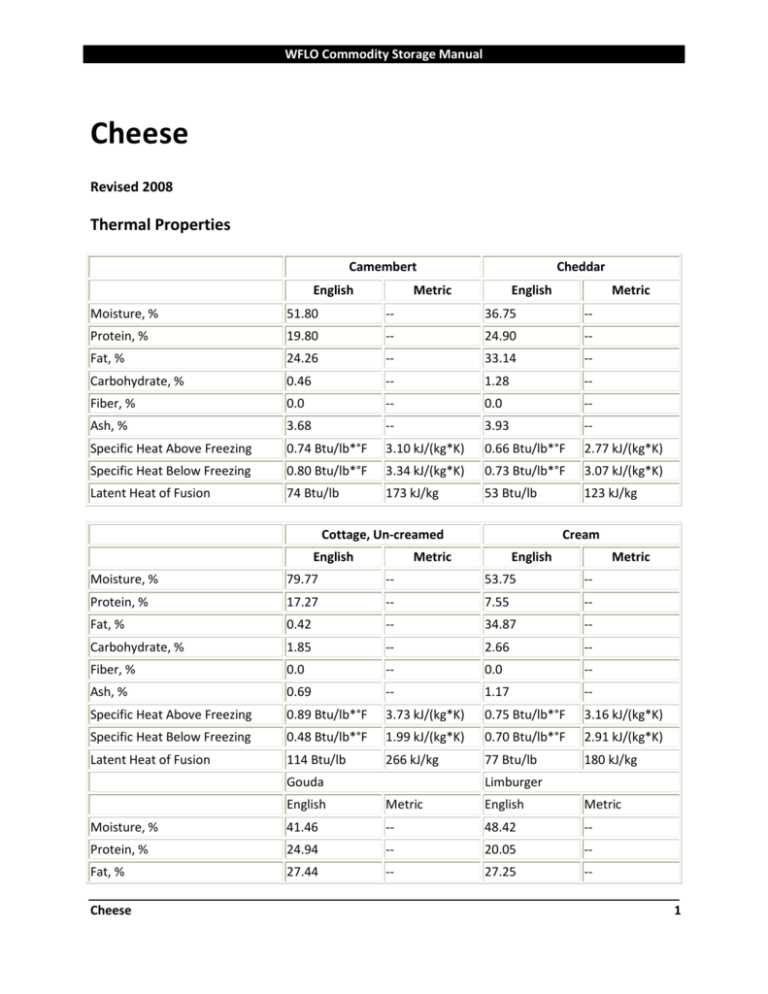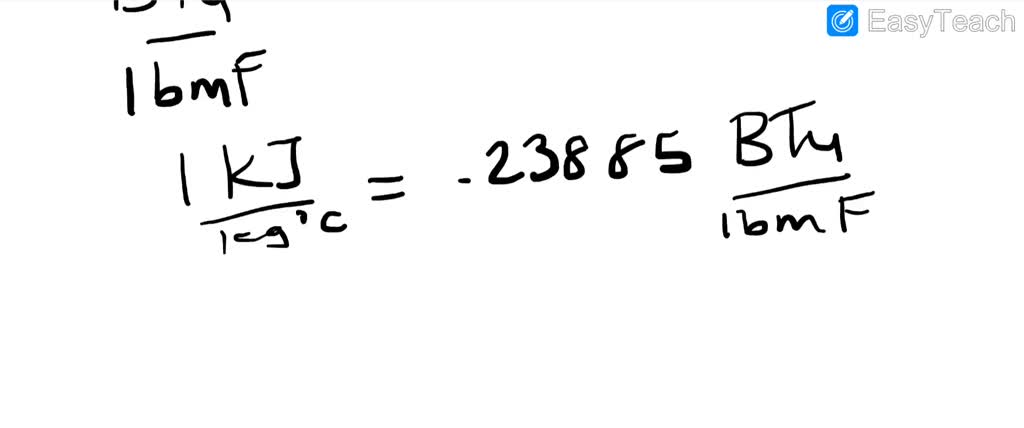
SOLVED:Specific heat is defined as the amount of energy needed to increase the temperature of a unit mass of a substance by one degree. The specific heat of water at room temperature

Units conversion - Conversion Factors Mass and Density Pressure 1 1 1 1 1 1 1 Pa kg g/cm3 g/cm3 lb - Studocu