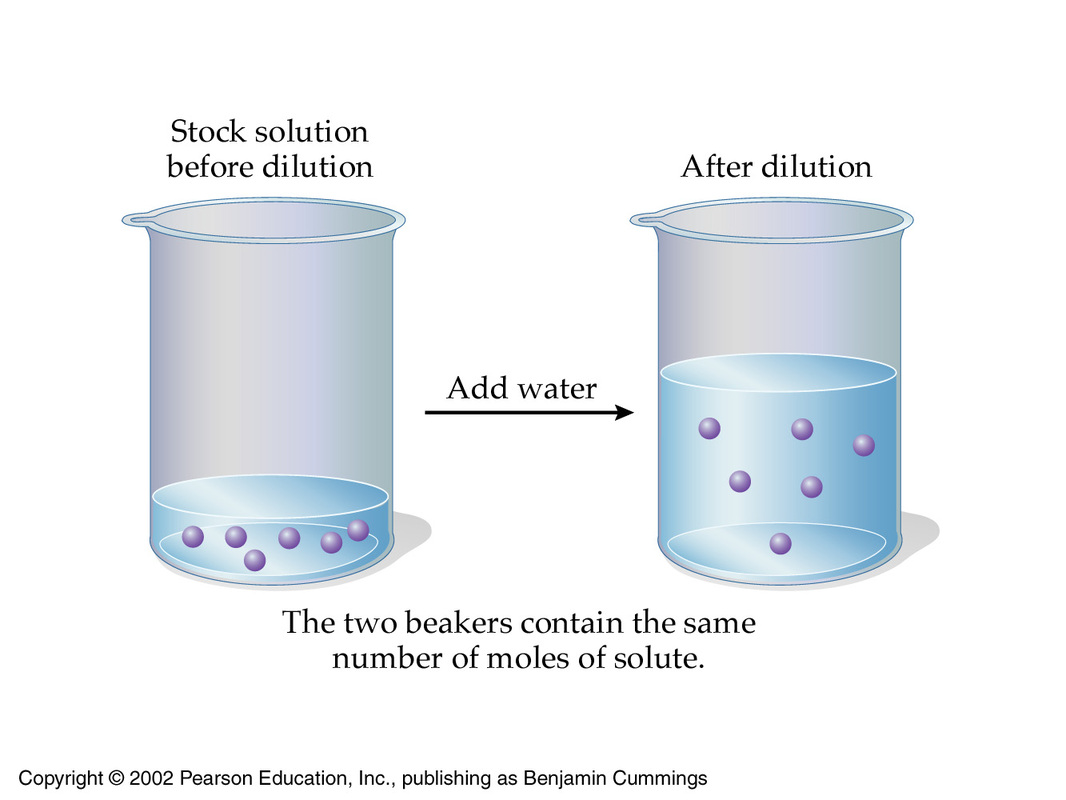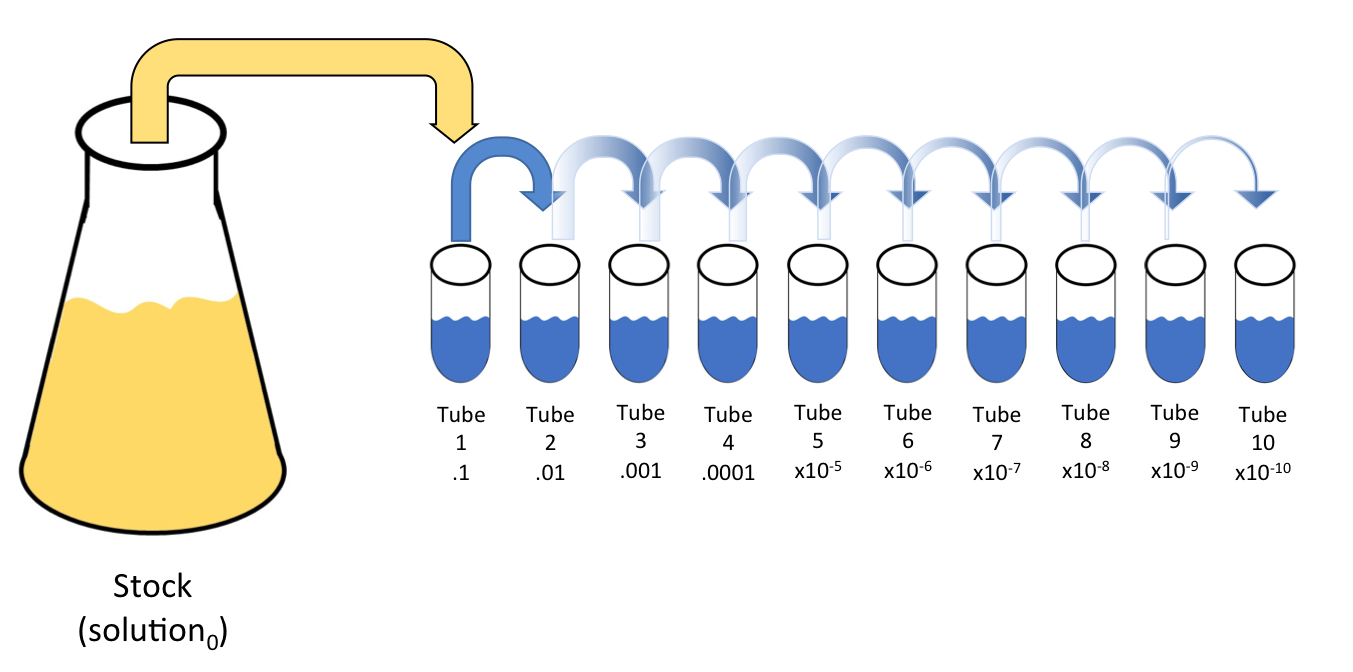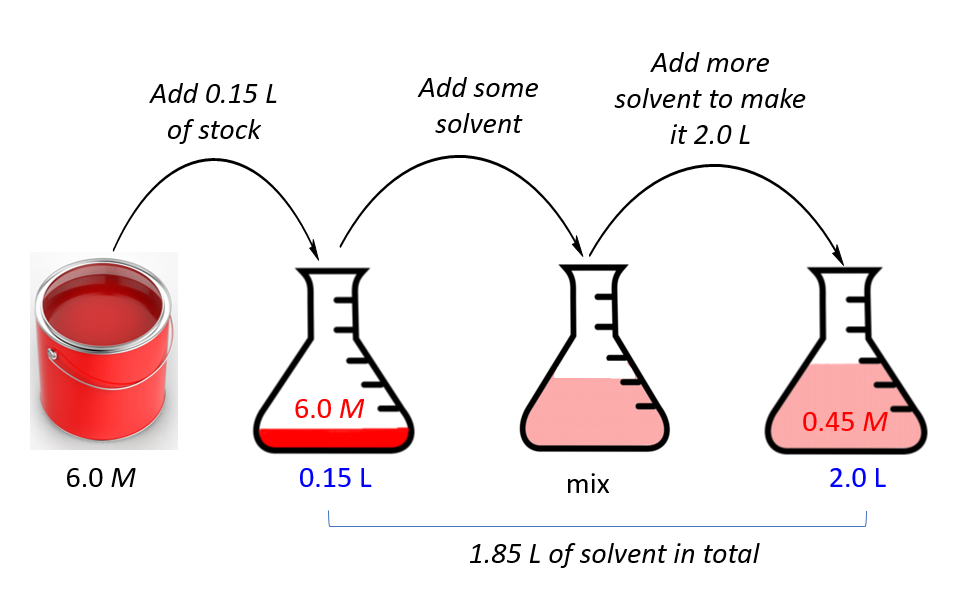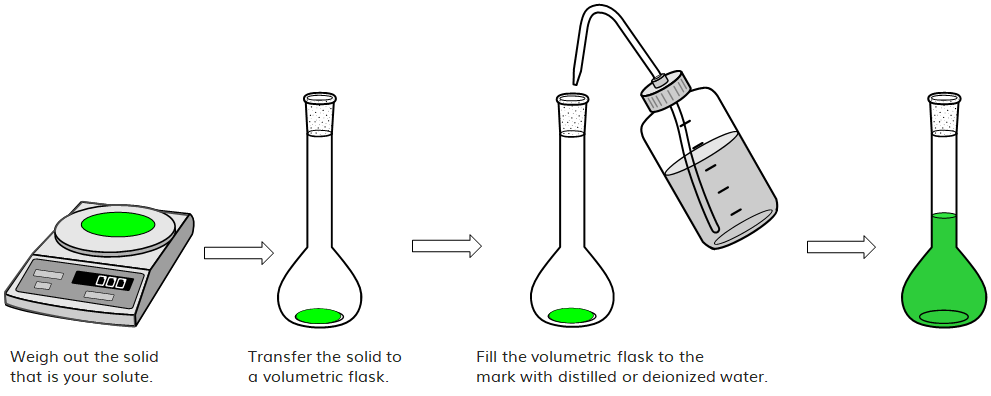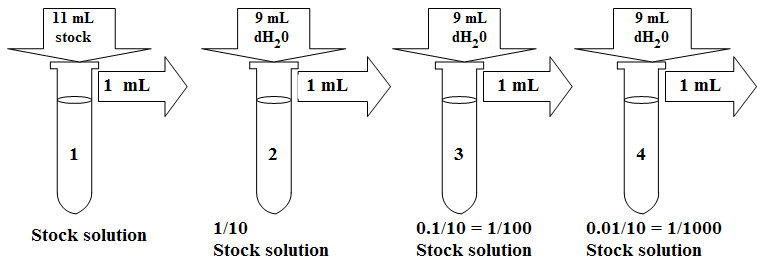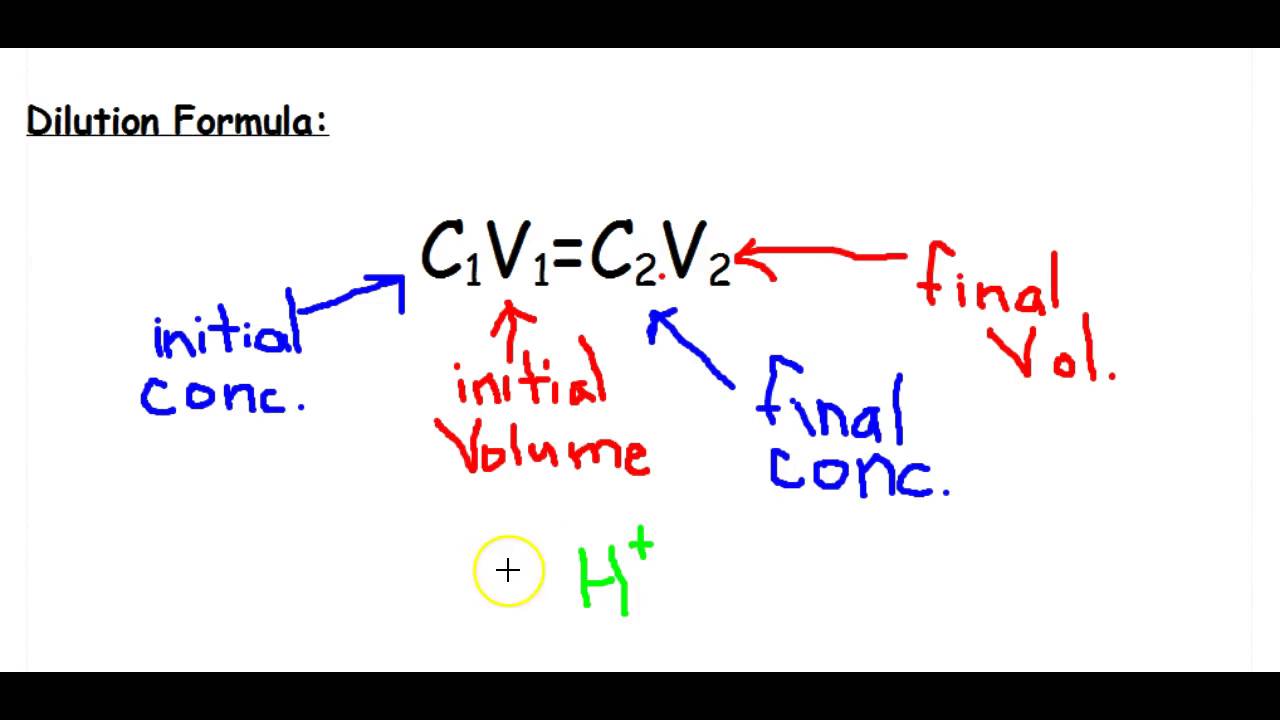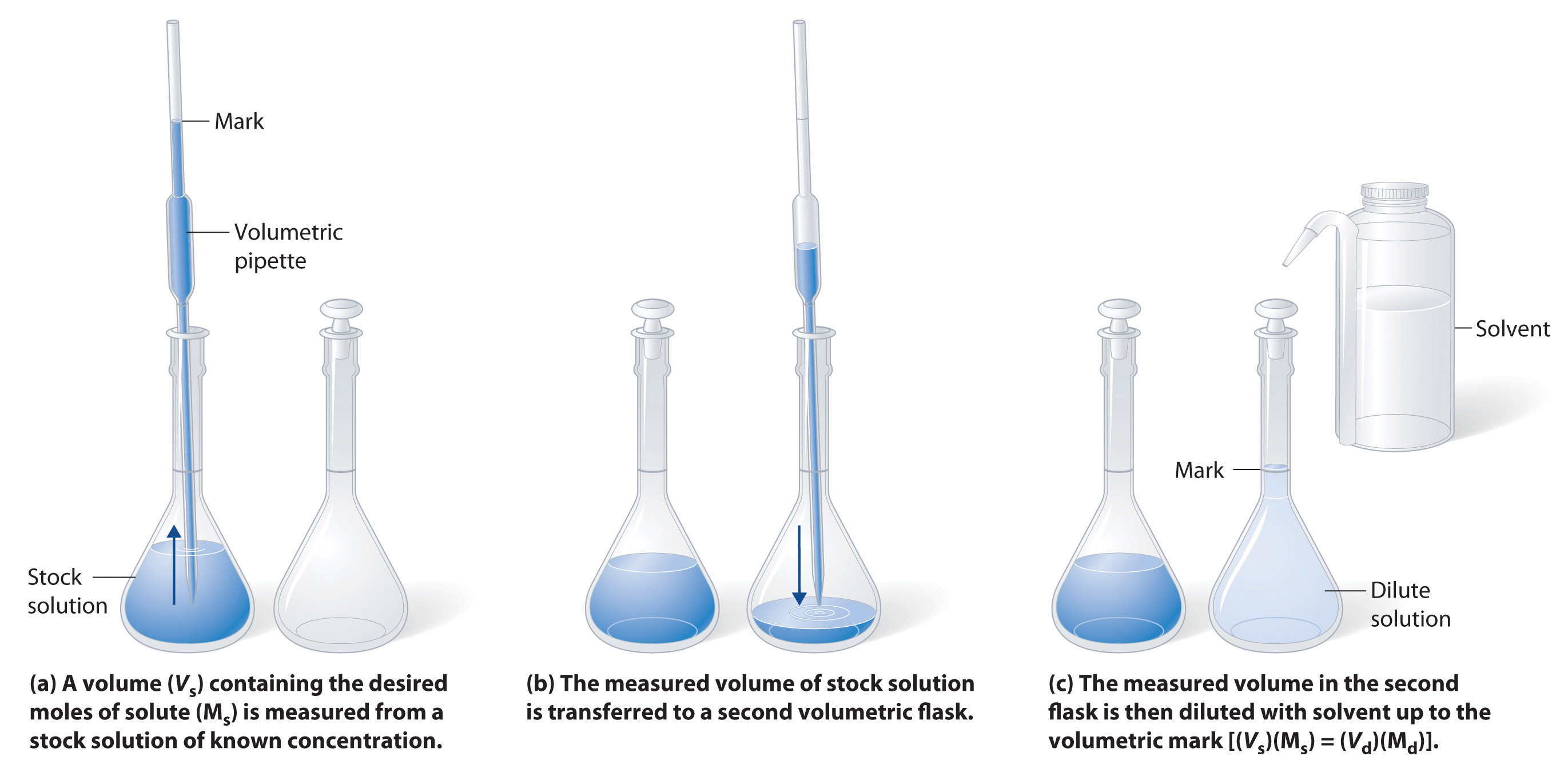
What volume (in mL's) of a 6.0 M stock solution of HCl should be used to prepare 6.00 L of a 0.250 M HCl solution? | Socratic

Titration of puromycin. The stock solution of puromycin (10 mg/mL) is... | Download Scientific Diagram

Difference Between Stock Solution and Standard Solution | Compare the Difference Between Similar Terms